Introduction – What Is Phenolphthalein?
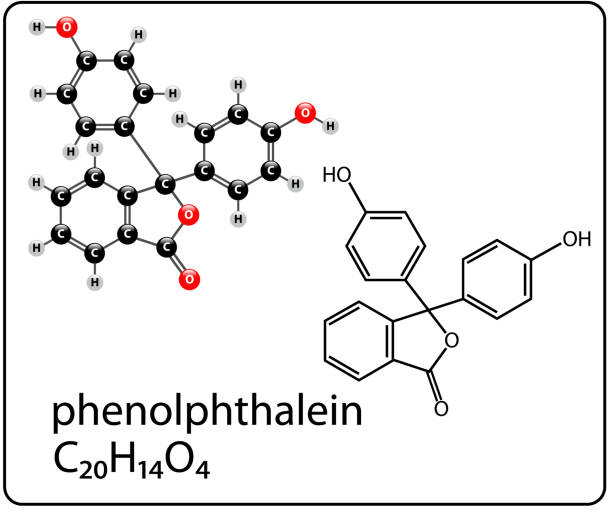
Phenolphthalein CAS 77-09-8 is a classic acid-base indicator and pH indicator widely used in both laboratory and industrial analyses. Its molecular formula is C20H14O4, and its structure comprises aromatic rings linked to a lactone (phthalide) framework. In acidic solutions, phenolphthalein exists in a colorless form; in basic media, it shifts to pink or fuchsia, providing a clear visual cue for endpoint detection in titrations and other analytical procedures.
Phenolphthalein serves as a fundamental acid-base indicator in titrations and qualitative analyses. It is valued for its sharp color transition in the alkaline region, which helps analysts determine endpoint conditions accurately. Beyond the classroom, phenolphthalein plays a critical role in various industrial processes where pH monitoring and control are essential for product quality and process safety.
Chemical Structure and Physical–Chemical Properties
Overview of structure
Phenolphthalein features two benzene rings connected through a central phthalide lactone moiety. This arrangement enables its pH-dependent structural changes that drive color transitions.
The compound is commonly described by its abbreviated labels HIn for the neutral/acid form and In− for the deprotonated form that contributes to the characteristic color change in alkaline solutions.
Physical properties
- Appearance: White to pale pink crystalline powder, depending on purity and particle size.
- Melting point: Approximately 258–263 °C, indicating good thermal stability for handling and storage in powder form.
- Solubility: Practically insoluble in water; soluble in alcohols (e.g., ethanol, isopropanol), which is why solutions for titration are typically prepared in alcoholic vehicles or with co-solvents.
Chemical behavior and indicator range
- Acidic solutions: Colorless form predominates.
- Basic solutions: Turns pink to fuchsia as the pH increases, establishing its use as a pH indicator in the basic region.
- pH transition range: Roughly pH 8.2 to 10.0, with the most vivid color change occurring around pH 8.3–10.0, depending on concentration and solvent system.
Practical note: At very high pH levels or under strong alkaline conditions, the color may fade or shift in intensity due to further structural changes; in typical analytical contexts, the sharp color transition near the neutral-to-weakly-alkaline region is exploited for clear endpoint definition.
How Phenolphthalein Works as an Indicator
Phenolphthalein acts as an acid-base indicator whose color is dictated by the molecule’s structural state, which changes with pH. In acidic conditions, the molecule exists primarily in a colorless lactone form, while at higher pH, the structure opens and extended conjugation produces a vivid pink to fuchsia color. This color-shift behavior is the core reason why phenolphthalein is widely used in titrations and analytical procedures.
Molecular basis and diagnostic range
The indicator’s color change arises from distinct resonance structures: the non-ionized form in acidic media is colorless, while deprotonated forms in basic media extend the conjugation, producing pink to magenta hues. As pH increases beyond the neutral region, the signal can shift in intensity depending on solvent and concentration, but the characteristic alkaline color is robust around the endpoint region.
Typical pH range for a sharp transition is approximately 8.2 to 10.0, with the most vivid coloration occurring near pH 8.3–10.0 under standard conditions. Variations in the solvent system or concentration can slightly shift this range, a consideration for method development.
For routine pH monitoring in manufacturing and quality-control workflows, phenolphthalein can function as a quick-check indicator to confirm alkaline conditions or to verify endpoint completeness in automated processes. Its solid colorimetric response and straightforward handling make it a practical choice in many industrial settings.
Industrial and Laboratory Applications of Phenolphthalein
Laboratory applications
Acid-base titrations: Used to determine endpoint pH in titrations where the equivalence point lies in the alkaline region. A small amount of indicator is added to the analyte; titration proceeds until the persistent pink color indicates the endpoint. This straightforward visual cue simplifies qualitative analyses and helps ensure repeatable measurements.
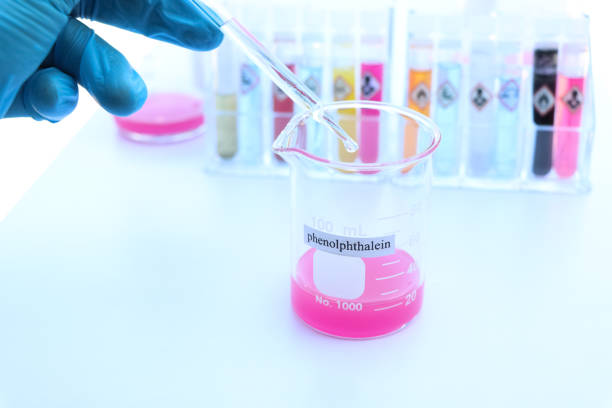
Industrial applications
Chemical manufacturing and process control: Phenolphthalein serves as a pH-monitoring tool to adjust neutralization steps, maintain reactor conditions, and verify post-treatment acidity/alkalinity. Its clear color transition supports timely interventions in production lines.
Water treatment and environmental testing: Used to assess alkalinity and to monitor neutralization in discharge streams, helping facilities meet regulatory standards and optimize chemical usage for environmental compliance.
Pharmaceutical and quality control: In manufacturing settings, phenolphthalein is employed to verify pH specifications of formulations, inerting processes, and raw materials before downstream processing. It supports consistent product quality and traceability in QA/QC workflows.
Educational and research relevance
Demonstration and method development: The distinct color change provides a clear visual aid for teaching acid-base concepts, calibrating instruments, and validating new analytical methods in research settings. It remains a baseline indicator for developing and illustrating titration techniques.
Advantages of Using Phenolphthalein Indicator
Observability and sensitivity
- Distinct color change: The switch from colorless in acidic media to pink-to-magenta in basic media is broad and easy to notice, reducing observer error and improving reproducibility across operators.
- Suitable for teaching and demonstration: The dramatic color shift provides a highly effective visual aid for illustrating acid-base concepts, titration endpoints, and indicator theory in classrooms and training environments.
Cost and stability
- Cost-effective: Phenolphthalein is widely available at low cost, enabling routine use in high-throughput analyses and large-scale industrial QC without compromising performance.
- Stability: When stored properly (dry, sealed, away from moisture and strong oxidizers), phenolphthalein powder and prepared solutions demonstrate reliable shelf-life suitable for long-term procurement programs.
Industrial and regulatory relevance
- Process control and QA/QC: Clear endpoint signaling supports consistent product quality in manufacturing processes requiring pH adjustments, neutralization steps, and routine monitoring.
- Regulatory alignment: The well-established use of phenolphthalein in validated analytical methods makes it a familiar, auditable component in quality systems across pharmaceutical, chemical, and environmental testing sectors.

Comparison with other indicators
- Methyl orange: Effective for strong acid–strong base titrations, but changes color in the acidic region, which can limit visibility and endpoint precision for alkaline titrations. Phenolphthalein excels in alkaline-range titrations due to its sharp transition in that region.
- Bromothymol blue (phenol red family): Has a broader pH range and color transitions that may complicate endpoint identification in some titrations; phenolphthalein’s transition near the alkaline end provides a clear, high-contrast endpoint for many standard procedures.
Safety Information and Handling (Phenolphthalein MSDS)
MSDS Key Points Summary
Historically, phenolphthalein has been classified as a suspected carcinogen (although its use in indicators is minimal). The main risks associated with the use of modern industrial and laboratory-grade phenolphthalein are its irritation to the eyes and skin. Inhalation of its powder or vapor may irritate the respiratory tract.
Exposure Protection
- Skin and Body: Wear lab coats and chemically resistant gloves (such as nitrile rubber gloves) during handling.
- Eyes: Safety goggles or a face shield must be worn to prevent powder or solution splashes.
- Respiratory Tract: In areas where powder is dispersed or in poorly ventilated environments, local ventilation or a qualified dust mask may be necessary.
First Aid
- Skin Contact: Immediately wash contaminated skin with plenty of soap and water.
- Eye Contact: Immediately flush eyes with plenty of water for at least 15 minutes and seek medical attention.
- Inhalation: Move quickly to fresh air. If breathing is difficult, perform artificial respiration and seek medical attention.
Storage and handling precautions
- Storage Environment: Store in a cool, dry, well-ventilated area, away from incompatible substances and strong oxidizers.
- Containers: Keep containers tightly closed to prevent moisture and light exposure.
- Operating Area: Weighing and solution preparation should be carried out in a fume hood to minimize powder inhalation.
- Waste Disposal: Any waste containing phenolphthalein must be sorted and safely disposed of in accordance with local and national chemical waste management regulations. Never pour high-concentration solutions directly down the drain.
Why Choose Autech Industry as Your Phenolphthalein Powder Supplier

- High Purity and Stability: Autech Industry provides high-purity Phenolphthalein powder, ensuring endpoint repeatability and data traceability. Its excellent stability makes it suitable for large-volume purchasing and long-term inventory management.
- Reliable Supply Capability: Autech Industry possesses stable production lines, flexible inventory strategies, and cross-regional logistics capabilities to meet the needs of various sizes, from educational institutions to large manufacturing enterprises.
- Complete Packaging Options: We offer a variety of packaging options (such as small packages, container loading, ACS/USP level labeling, etc.) for rapid deployment in different scenarios.
- Technical and Compliance Support: We provide detailed technical data and safety documentation, including accompanying MSDS/SDS, usage instructions, and storage and usage recommendations, to help customers quickly complete internal approvals and training.
Conclusion
Phenolphthalein CAS 77-09-8 is a classic and reliable acid–base indicator widely used in laboratories and industrial analysis. With its clear color change and stable chemical behavior, it plays an essential role in titration and pH testing. Beyond its indicator function, phenolphthalein also has value in research, quality control, and chemical manufacturing.
Autech Industry provides high-purity phenolphthalein powder with consistent quality, complete MSDS documentation, and flexible packaging options. Contact us today to learn more about our phenolphthalein chemical and how we can support your laboratory or industrial needs.
FAQs About Phenolphthalein
1. What is the chemical formula of Phenolphthalein?
The chemical formula of phenolphthalein is C₂₀H₁₄O₄. It is an organic compound that serves as a well-known acid–base indicator in chemistry.
2. Why does Phenolphthalein change color?
Phenolphthalein changes color because of its molecular structure transformation in different pH environments. It is colorless in acidic solution and turns pink to fuchsia in basic solution (around pH 8.2–10.0). This makes it ideal for acid–base titrations and pH testing.
3. Is Phenolphthalein safe to use in laboratories?
Yes, phenolphthalein is generally safe when handled properly. However, users should follow the MSDS safety guidelines, avoid ingestion or inhalation, and wear protective gloves and goggles during handling. Autech Industry provides phenolphthalein with full MSDS documentation and packaging compliant with international safety standards.
4. Where can I buy Phenolphthalein powder in bulk?
You can purchase high-purity phenolphthalein powder directly from Autech Industry, a reliable supplier of fine chemical products. We offer bulk quantities, prompt shipment, and professional support for laboratory and industrial clients worldwide.